COVID-19 VACCINATION / THERAPEUTIC INFORMATION
Board staff have consolidated COVID-19 vaccine related items posted on our homepage and organized them below by topic. Select a topic to be taken to that section:
1) Vaccine / Therapeutics Administration Authorization (PAGE UPDATED OCTOBER 6, 2023)
2) NC DHHS's COVID-19 Vaccine Management System (CVMS) Provider Enrollment Portal
4) Other Vaccine Related Resources for Pharmacy Practitioners

NC DHHS COVID-19 VACCINATION INFORMATION FOR PROVIDERS: https://covid19.ncdhhs.gov/guidance#vaccination-info-for-providers.
NC STATE HEALTH DIRECTOR STANDING ORDERS FOUND HERE.
1) VACCINE ADMINISTRATION AUTHORIZATION
FROM THE NC DEPARTMENT OF HEALTH AND HUMAN SERVICES - SEPTEMBER 13, 2023
Health Officials Encourage North Carolinians to Get Fall COVID-19 Vaccine and Flu Shot.
NC STATE HEALTH DIRECTOR STANDING ORDERS FOUND HERE.
THURSDAY, SEPTEMBER 7, 2023
NC DHHS ISSUES UPDATED COVID-19 TREATMENT GUIDANCE. COVID-19 cases are rising in North Carolina. NC DHHS has issued updated treatment guidance to health care providers, found here.
FRIDAY, SEPTEMBER 1, 2023
FROM THE NC DEPARTMENT OF HEALTH AND HUMAN SERVICES: IMPORTANT IMMUNIZATION UPDATES - COVID-19 VACCINE COMMERCIALIZATION PLAN AND RSV MONOCLONAL ANTIBODY.
COVID-19 Vaccine Commercialization
FDA authorization and/or approval and the Advisory Committee on Immunization Practices (ACIP)/Centers for Disease Control and Prevention (CDC) recommendation of the updated, monovalent (XBB 1.5) 2023-2024 COVID-19 vaccine is expected the week of September 11, 2023. ACIP is scheduled to vote on the 2023-2024 COVID-19 vaccine on September 12, 2023.
Once ACIP recommends the updated COVID-19 vaccines and CDC’s director approves, the current bivalent COVID-19 vaccines will no longer be authorized for use. At that time, you will be required to stop administering the bivalent doses and waste any remaining doses on hand following your agency’s disposal policies as well as appropriately waste inventory in NCIR. Administration of non-authorized products is considered a vaccine administration error and must be reported to the Vaccine Adverse Events Reporting System (VAERS).
Follow this link for more detailed information.
WEDNESDAY, JULY 19, 2023
UPDATED FAQ GUIDANCE ON PHARMACIST AND PHARMACY TECHNICIAN VACCINATION AUTHORITY. Board staff have updated the FAQ guidance document on pharmacist and pharmacy technician vaccination authority. This guidance details state-law vaccination authority and how that authority intersects with two other sources of authority – declarations under the federal PREP Act and the State Health Director’s standing orders for pharmacist-administered COVID-19 vaccines.
2) NC DEPARTMENT OF HEALTH & HUMAN SERVICES COVID-19 VACCINE MANAGEMENT SYSTEM (CVMS) PROVIDER ENROLLMENT PORTAL
FRIDAY, JANUARY 15, 2021
FROM THE NC DEPARTMENT OF HEALTH & HUMAN SERVICES: COVID-19 VACCINE MANAGEMENT SYSTEM (CVMS) PROVIDER ENROLLMENT PORTAL
We have received an influx of applications since the COVID-19 Vaccine Management System (CVMS) Provider Enrollment Portal opened to providers on January 11, 2021 and thank you for your enthusiasm around COVID-19 vaccine administration. Given the number of applicants, we want to provide insight into how providers are currently being approved for enrollment.
Please understand that while qualified providers are welcome to enroll, applications will be reviewed in an order aligned with the prioritization phases, and the timeline for approval will vary. The table below highlights the six prioritization groups as well as target timing for enrollment decisions.
Provider Type |
Target Approval Timing |
(1) Hospitals, Health Systems, LHDs |
November / December |
(2) FQHCs, LTC Pharmacies, LHD Identified Partners, Correction Health, Mass Vaccinators |
December / January |
(3) Family Medicine/Internal Medicine, Medical Directors SNF, Occupational Health |
January / February |
(4) Retail Pharmacy, Pediatric Primary Care, Independent Practice Specialty |
February / March |
(5) Other Providers (e.g. Dental, Optometry) |
TBD pending executive authorization to administer COVID-19 vaccines |
(6) Other Entities (e.g. Labs, Colleges, Universities) |
TBD |
We ask for your patience as we review the thousands of provider applications. Please also remember that provider enrollment approval does not guarantee vaccine allocation. We will update you as we receive more information on the timing and prioritization of enrollment applications.
Thank you for your continued support,
NC Immunization Branch
MONDAY, JANUARY 11, 2021
From the NC Department of Health and Human Services:
We are writing to inform you that starting today (1/11/21), North Carolina’s COVID-19 Vaccine Management System (CVMS) Provider Enrollment Portal is open. Please share this message with any providers you are aware are eligible and interested in administering the COVID-19 vaccine.
Any provider enrolling through the portal will be reviewed and considered for approval by the NC Immunization Branch to administer the COVID-19 vaccine. Enrollment can be initiated here: https://covid-enroll.ncdhhs.gov/s/login/?ec=302&startURL=%2Fs%2F.
As a reminder, COVID-19 vaccine providers must be qualified under the CDC agreement to prescribe COVID-19 vaccines and authorized under the appropriate NC licensing authority. Enrolled COVID-19 vaccine providers must be credentialed/licensed in North Carolina. Please understand that while currently qualified providers are welcome to enroll, requests will be addressed in an order aligned with the prioritization phases and the timeline for approval will vary. The State will continuously reassess enrollment prioritization based on needs.
Please visit the North Carolina Immunization Branch COVID-19 training website for Provider Enrollment Portal training content: https://immunize.nc.gov/providers/covid-19training.htm. The available modules are captured in the screenshot below.

Additional trainings related to CVMS can also be found on https://immunize.nc.gov/providers/covid-19training.htm. Please note, any trainings you elect to review at this time are subject to be refreshed by the time you become an approved provider. You may need to revisit trainings depending on approval timelines.Thank you for your continued support,
NC Immunization Branch
3) COVID-19 VACCINATION TRAINING OPPORTUNITIES FOR PHARMACISTS AND TECHNICIANS
Free Monoclonal Antibody Assessment & Administration CE. APhA has released a new and free training program to help meet the 9th Amendment of the PREP Act requirements! Coupled with the APhA Immunization Certificate, these activities meet the requirements of all PREP Act amendments thus far to order and administer COVID-19 Immunizations and Therapeutics.
The training program covers the clinical evaluation of indications and contraindications of COVID-19 therapeutics, adverse events, and the specifics related to the FDA approval, authorization, clearance, or licensing of COVID-19 therapeutics. You can find the training here: COVID-19 Monoclonal Antibody Assessment & Administration.
****************************************
COVID-19 Vaccination in North Carolina Parts 1 and 2 on-demand videos are now available on our website https://cmetracker.net/CAMPBELL/Catalog
Follow the instructions provided at registration to view the videos. Be sure to record the activity code and claim credit at the following link.
HOW TO CLAIM CE CREDIT:
The activity code to claim credit is provided at the end of the presentation. Record the code and sign in here: https://cmetracker.net/CAMPBELL/Catalog
Credit Instructions: https://cmetracker.net/CAMPBELL/Files/EventMaterials/3239.pdf
Slides: https://cmetracker.net/CAMPBELL/Files/EventMaterials/3211.pdfContact: Samantha Clinton, CPhT, CQEP, HMCC
Assistant Director of Continuing Professional Education
Campbell University College of Pharmacy & Health Sciences
910.814.4784 | clintons@campbell.edu
Description: This two-part continuing pharmacy education course has been developed in partnership with the North Carolina Board of Pharmacy to provide education and guidance for pharmacists and pharmacy technicians on the novel COVID-19 virus, COVID- 19 vaccines in development and currently available, COVID-19 vaccine distribution plans in North Carolina, vaccine administration strategy, and federal and state regulatory issues governing pharmacist, pharmacy intern, and pharmacy technician administration of COVID-19 vaccines.
*************************************
4) OTHER VACCINE RELATED TOPICS AND RESOURCES FOR PRACTITIONERS
WEDNESDAY, SEPTEMBER 15, 2021
FDA, CDC, AND NC DHHS CONTINUE TO WARN AGAINST THE USE OF IVERMECTIN FOR PREVENTION OR TREATMENT OF COVID-19. Pharmacists have reported receiving increased numbers of prescriptions for ivermectin, as well as hostile and abusive communications concerning the same from some prescribers and patients.
The United States Food and Drug Administration has re-emphasized that ivermectin is not approved for the treatment or prevention of COVID-19. It is approved for the treatment of intestinal conditions caused by certain parasitic worms and, in topical formulation, certain external parasites. Veterinary formulations are approved to treat certain internal and external parasites – but not to treat any condition in humans. FDA emphasizes that taking large doses of ivermectin is dangerous. And no reliable studies support the safety or efficacy of ivermectin in the treatment or prevention of COVID-19. More information from FDA is found here: https://www.fda.gov/consumers/consumer-updates/why-you-should-not-use-ivermectin-treat-or-prevent-covid-19.
The United States Centers for Disease Control issued an alert on August 26, 2021 noting a sharp rise in serious illness associated with ingestion of ivermectin products (human and veterinary) for purported treatment or prevention of COVID-19. Severe gastrointestinal and neurotoxic effects are being reported. Like the FDA, the CDC emphasizes that ivermectin is not approved for the treatment or prevention of COVID-19, no reliable studies support its use in the treatment or prevention of COVID-19, and improper use is dangerous. More information from CDC is found here: https://emergency.cdc.gov/han/2021/han00449.asp.
State Health Director Betsey Tilson has also issued a warning in light of spike in prescriptions, poison control calls, and emergency room visits. Dr. Tilson likewise reminds health care practitioners that ivermectin is neither approved nor authorized by FDA to treat or prevent COVID-19 – and that is not a substitute for COVID-19 vaccine or approved treatments for the disease. Dr. Tilson asks that all providers educate patients about the dangers of using ivermectin: http://www.ncbop.org/PDF/COVID19NCDHHSIvermectinRisksSept2021.pdf.
The North Carolina Medical Board has communicated these warnings to its licensees: https://www.ncmedboard.org/resources-information/professional-resources/publications/forum-newsletter/notice/cdc-warns-against-inappropriate-ivermectin-use. And the Federation of State Medical Boards has warned physicians that spreading COVID-19 misinformation risk disciplinary action by state medical boards, including suspension or revocation of license. https://www.fsmb.org/advocacy/news-releases/fsmb-spreading-covid-19-vaccine-misinformation-may-put-medical-license-at-risk/
Pharmacists are reminded that they have a right to refuse to fill or refill any prescription order if doing so would be contrary to their professional judgment. Moreover, a pharmacist shall not fill or refill a prescription order if, in the exercise of the pharmacist’s professional judgement, there is a question as to its safety for the patient. 21 NCAC 46.1801. As well, pharmacists have an obligation to engage in neither negligent nor unprofessional conduct in the practice of pharmacy. G.S. 90-85.38(a)(9) and (a)(10).
WEDNESDAY, JANUARY 27, 2021
PHARMACY TECHNICIANS QUALIFIED TO ADMINISTER VACCINES UNDER THE FEDERAL PREP ACT SHOULD UPDATE THEIR STATUS THROUGH THE BOARD’S LICENSING PORTAL. The U.S. Department of Health and Human Services has issued a declaration under the PREP Act authorizing qualified pharmacy technicians to administer ACIP-recommended pediatric vaccines and COVID-19 vaccines under the supervision of a qualified pharmacist: http://www.ncbop.org/PDF/PREPActTechnicianVaccination102220.pdf Technicians who have completed the required training to administer vaccines under this declaration should log on to their profile through the Board’s Licensing Portal and update their status as vaccinators.
Once logged in to the technician’s profile, click on the “Pharmacy Technician” tile, then a “Vaccinator” tile will appear on the next page. A technician now qualified to administer vaccines should hover over that tile, note the attestation, and then click to turn the tile green and indicate vaccinator status.
Click on the Pharmacy Technician tile:
Then, click on the vaccinator tile to indicate a technician is vaccinating:
WEDNESDAY, NOVEMBER 18, 2020
IMMUNIZING PHARMACIST CONTINUING EDUCATION REQUIREMENTS UNDER THE US DHHS PREP ACT DECLARATION VERSUS REQUIREMENTS UNDER NORTH CAROLINA LAW. As pharmacists know, the US DHHS has issued declarations under the PREP Act authorizing pharmacists to order and administer ACIP-recommended pediatric vaccines and COVID vaccines: http://www.ncbop.org/PDF/COVID19PREPActVaccinationAug2020.pdf; http://www.ncbop.org/PDF/PREPActCOVIDVaccination090920.pdf
Board staff have received questions about the immunizing pharmacist CE requirements under the federal authorization versus CE requirements under North Carolina law. Under the PREP Act declaration, the licensed pharmacist must complete a minimum of two hours of ACPE-approved, immunization-related continuing pharmacy education during each State licensing period (annually in North Carolina). This CE requirement differs somewhat from North Carolina law. North Carolina law requires an immunizing pharmacist to maintain “documentation of three hours of continuing education every two years, designed to maintain competency in the disease states, drugs, and vaccine administration.” G.S. §90-85.3(i1)(3). Any pharmacist exercising authority granted under the PREP Act must slightly increase his/her immunization-related CE and acquire two (2) hours in 2020 and two (2) hours in 2021 (assuming, as is likely, that the federally-declared COVID-19 public health emergency remains in effect into 2021).
More information about this requirement, and several others, is found in the two guidance documents linked above.
MONDAY, OCTOBER 12, 2020
PHARMACISTS UPDATING THEIR IMMUNIZER STATUS. Many Pharmacists are updating their immunizer status in order to administer vaccines.
Note: A pharmacist who completed an immunization certification course, but has not been administering vaccines does not need to retake the course. The immunizing pharmacist must have a current provider-level CPR certification prior to administering vaccines, must begin obtaining required biennial immunization-specific CE, and must obtain training to participate in North Carolina Immunization Registry as required by the immunizing pharmacist statute.
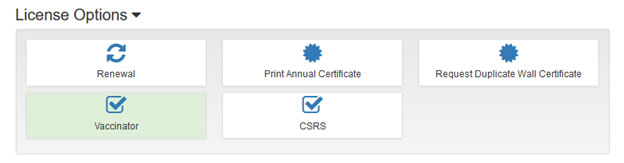
Here’s how to update the Board that you are a certified vaccinator:
- Log in on the Licensure Gateway page with your username and password
- Once logged in, scroll to the bottom of the page and click on the Pharmacist tile
- On the next page, under License Options, there is a tile for Vaccinator. If it’s white, then that means we don’t have you listed as a vaccinator in our database. If it’s green, then we do have you listed as a vaccinator. To change the color of the box, click on it.